Leading Center for Contract Research Organization
Two decades of excellence in pharmaceutical studies and solutions

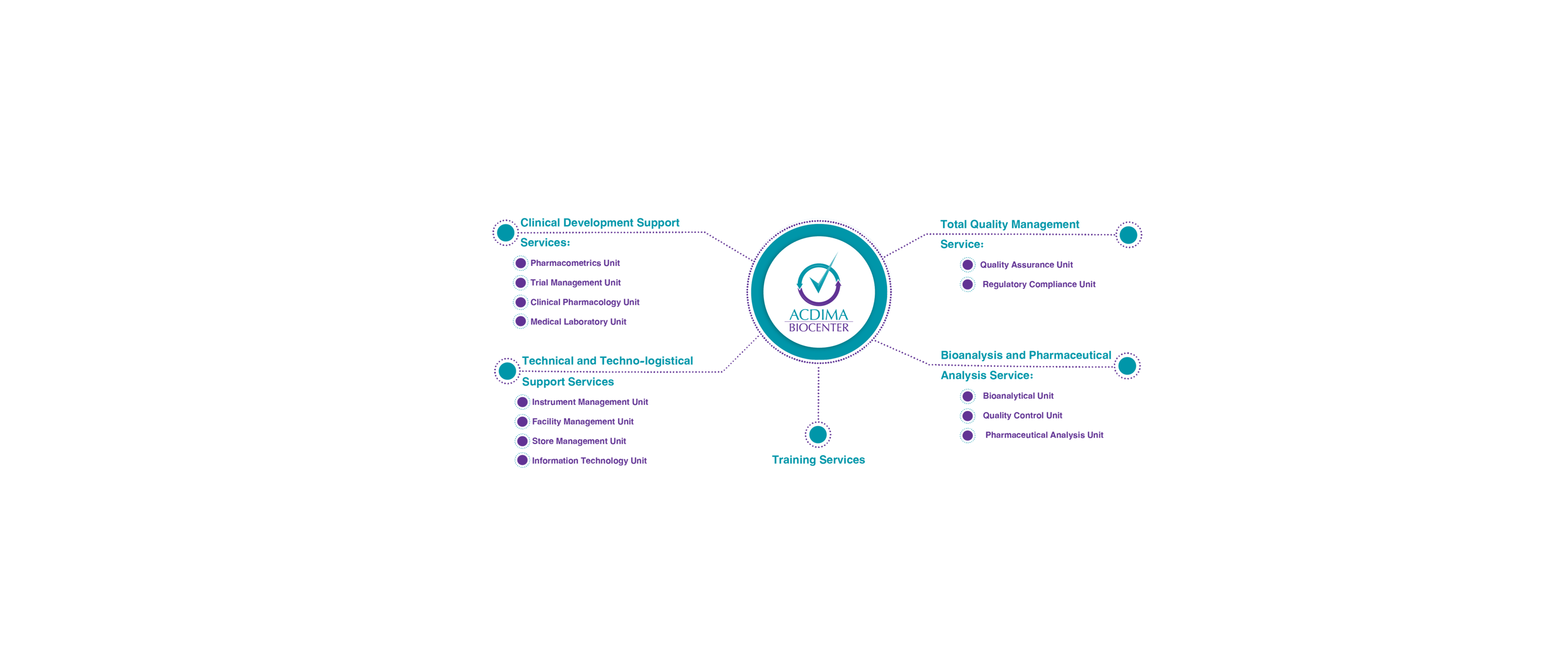
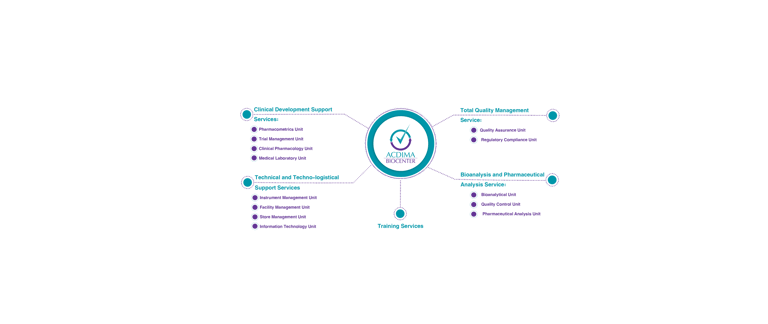
Clinical Development Support Services
Your trusted partner for streamlined clinical development


Bioanalysis and Pharmaceutical Analysis Services
Advancing science through innovative bioanalytical solutions

Technical and Techno-logistical Support Services
Streamlining your operations with our techno-logistical solutions
ACDIMA BioCenter has a strong track record of successfully passing multiple study-specific and system-based inspections conducted by the World Health Organization (WHO) under its Prequalification Programme, most recently in 2022. This reflects the Center’s continued commitment to compliance with applicable regulatory requirements, including Good Clinical Practice (GCP), Good Laboratory Practice (GLP), and other internationally recognized best practices.
Get in touch
Salah Al Shaimat St 18, Sweifieh, Amman, Jordan
Tel: + 962 6 582 1618
Fax: + 962 6 585 3719
P.O. Box: 925161 – Amman - 11190 - Hashemite Kingdom of Jordan
Employment
To apply for a job contact us, please send a cover letter together with your C.V. to: biocenter@acdima.com
Head Office
For any inquiries, questions or commendations, please call: + 962 6 582 1618 or fill out the following form
Excellence
Leading bioequivalence studies for global pharmaceutical solutions.
Quality
Tel: + 962 6 582 1618
© 2025. All rights reserved.